Keratoconus is a progressive eye disease that weakens and thins the cornea, the clear front surface of the eye. In its early, subclinical stage, the cornea can still appear normal on routine exams. Yet this is when accurate diagnosis matters most, especially when patients are being evaluated for refractive surgery. Recent research found that combining polarization-sensitive optical coherence tomography (PS‑OCT) with artificial intelligence reveals subtle corneal changes that standard imaging often misses. Published in Biophotonics Discovery, the study draws on a large clinical dataset and points to new ways to refine early keratoconus detection.
Most current screening tools rely on corneal shape. Devices such as Pentacam and MS‑39 measure curvature, thickness, and surface irregularities. These approaches work well once keratoconus is established, but are less reliable when disease-related changes are still microscopic. At that stage, the cornea may not yet show clear deformation, even though its internal structure is already changing. The authors therefore focused on a feature that shape-based imaging cannot capture directly: how collagen fibers inside the cornea are organized.
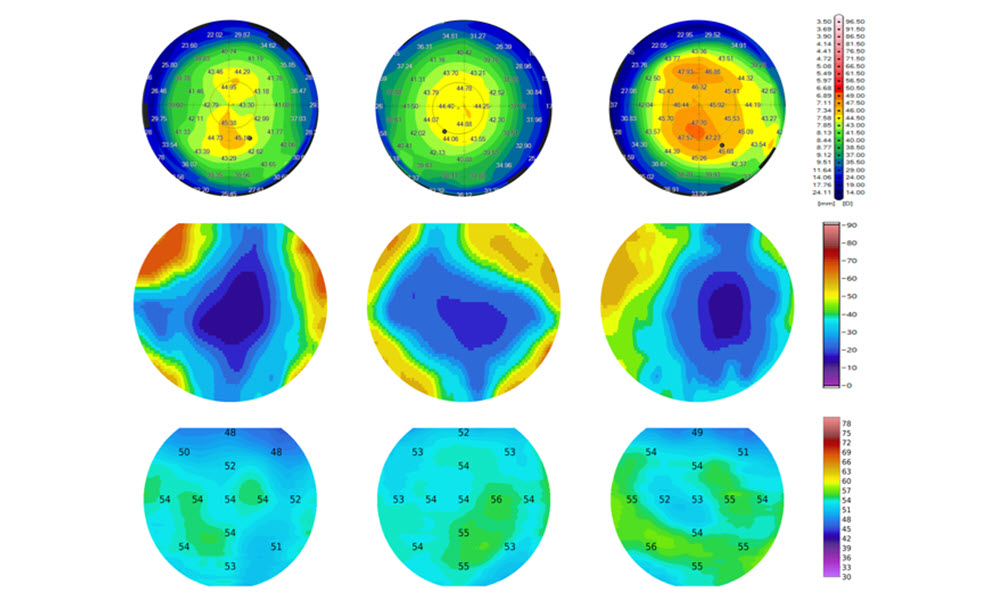
PS‑OCT is a high‑resolution imaging technique that detects how polarized light changes as it travels through tissue. In the cornea, these changes reflect the alignment of collagen fibers, which are essential for mechanical strength. Disruption of this collagen network is thought to occur early in keratoconus, before visible thinning or steepening develops.
In this study, the researchers used a custom-built PS‑OCT system capable of resolving fine corneal layers. From each scan, they derived maps of phase retardation, a measure related to collagen organization, along with thickness maps of three layers: the epithelium, Bowman’s layer, and the stroma.
The team analyzed imaging data from 359 eyes examined at Narayana Nethralaya Eye Hospital. The dataset included healthy eyes, eyes with clear keratoconus, and eyes classified as subclinical keratoconus based on established clinical criteria.
To compare diagnostic performance fairly, the authors trained three separate artificial intelligence models, each using data from a single device: PS‑OCT, Pentacam, or MS‑39. All models used the same machine learning method and cross‑validation strategy, allowing direct comparison of results.
For healthy eyes and for clear cases of keratoconus, all three models showed similar accuracy. Differences emerged in the subclinical group. Here, the PS‑OCT‑based model classified a larger fraction of eyes as healthy compared with the two tomography-based models.
Importantly, these reclassifications were not random. Eyes that PS‑OCT identified as truly subclinical showed higher phase retardation values and subtle differences in corneal sublayer thickness, even when overall corneal shape appeared normal. In contrast, eyes reclassified as healthy by PS‑OCT tended to have collagen and layer thickness patterns resembling those of normal corneas.
Detailed case examples highlighted these distinctions. In healthy eyes, phase retardation maps were uniform, and Bowman’s layer thickness was relatively even. Subclinical eyes showed modest but consistent increases in phase retardation in the central cornea, along with slight thinning of Bowman’s layer. Eyes with established keratoconus displayed highly irregular patterns across all maps.
These findings suggest that PS‑OCT is sensitive to microstructural differences that precede obvious shape changes. By capturing both structural and polarization-based information, the technique offers a different window into corneal health.
A common clinical dilemma is how to manage patients whose corneas are thin or slightly irregular but stable. Current methods often label such eyes as “suspect,” which can limit treatment options even when the risk of progression is low. The study shows that PS‑OCT can help distinguish true early disease from naturally thin but healthy corneas by focusing on collagen organization rather than thickness alone.
The authors emphasize that longer-term studies are needed to confirm whether eyes reclassified as healthy remain stable over time. Even so, their results indicate that polarization-sensitive imaging could improve confidence in early diagnosis and support safer decision-making in refractive surgery screening.
By combining ultrahigh-resolution imaging with artificial intelligence, this work highlights the value of looking beyond corneal shape. PS‑OCT does not replace existing tools but complements them by adding information about tissue integrity that standard tomography cannot provide. As imaging technologies continue to evolve, approaches like this may help clinicians detect keratoconus earlier and tailor care more precisely to individual patients.
For details, see the original Gold Open Access article by R. Patil et al., “Advancing subclinical keratoconus detection using polarization-sensitive optical coherence tomography and artificial intelligence," Biophoton. Discovery 3(1), 015004 (2026), doi 10.1117/1.BIOS.3.1.015004