In some atomic or molecular systems, light is absorbed by the system, undergoes some radiationless internal relaxation—either through vibrational states (vibrational relaxation), electronic states (internal conversion), or both—and then de-excites to the ground state by emitting a photon having a lower energy than the exciting photon. This process is called fluorescence.
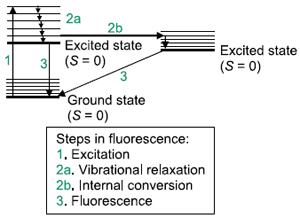
Fluorescence is a relatively fast process: 10-5-10-8 s. Fluorescence spectroscopy has three aspects: a plot of fluorescence versus wavelength of incident light; a plot of fluorescence versus the wavelength of emitted light; 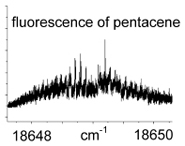 and the time frame in which the fluorescence is emitted. A diagram of the energy levels involved in a fluorescence process is called a Jablonski diagram. Because of the relaxation processes, fluorescence will usually be of a lower energy (i.e., lower frequency, higher wavelength) than the excitation light. High-resolution fluorescence spectra can show vibrational and, in the gas phase, rotational structure.
and the time frame in which the fluorescence is emitted. A diagram of the energy levels involved in a fluorescence process is called a Jablonski diagram. Because of the relaxation processes, fluorescence will usually be of a lower energy (i.e., lower frequency, higher wavelength) than the excitation light. High-resolution fluorescence spectra can show vibrational and, in the gas phase, rotational structure.
D. W. Ball, Field Guide to Spectroscopy, SPIE Press, Bellingham, WA (2006).
View SPIE terms of use.
